 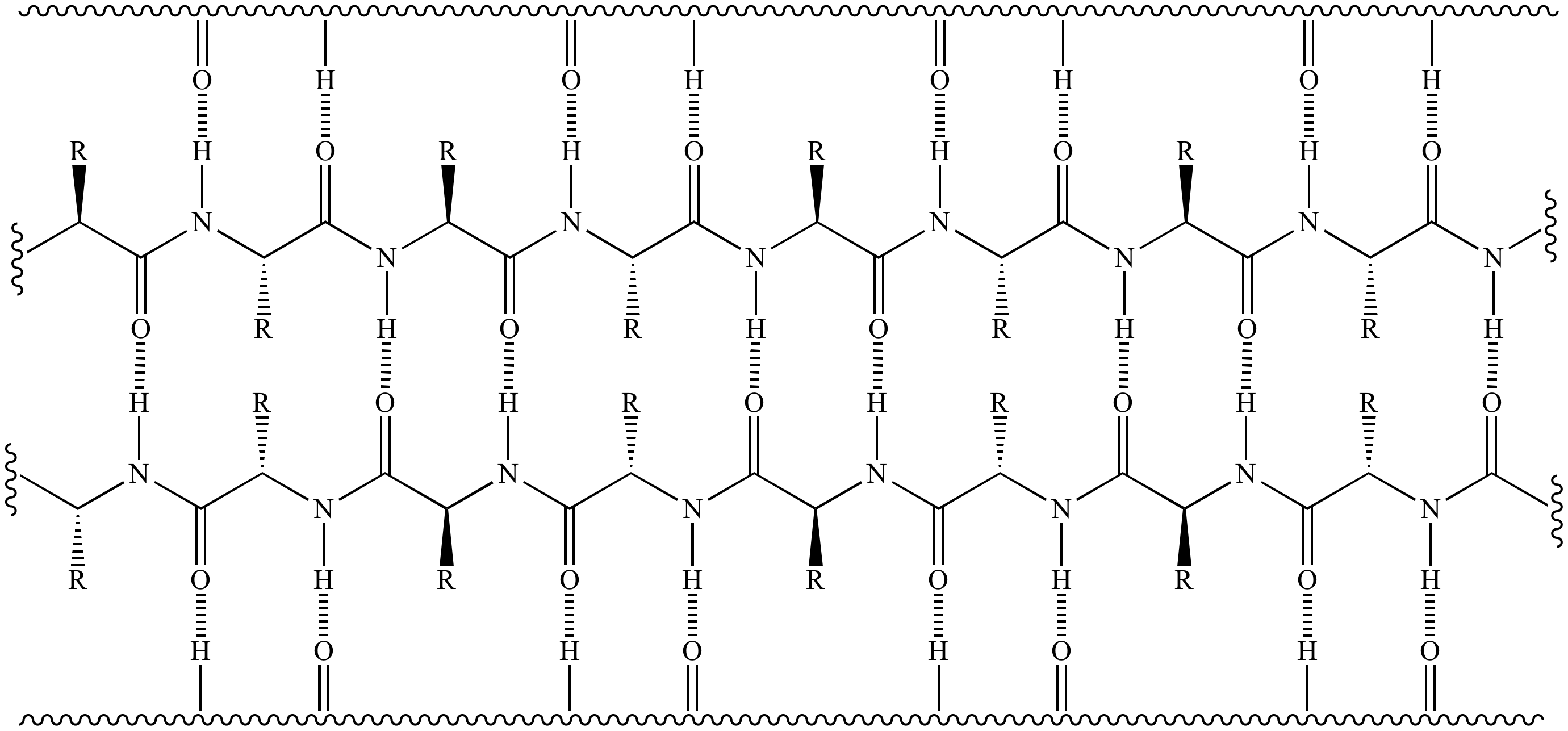
Although the importance of β-sheets in the folded structures of proteins has long been recognized, there is a growing recognition of the importance of intermolecular interactions among β-sheets. Β-Sheets consist of extended polypeptide strands (β-strands) connected by a network of hydrogen bonds and occur widely in proteins. Amino acids whose R-groups are too large (tryptophan, tyrosine) or too small (glycine) destabilize α-helices.“What I cannot create, I do not understand. The structural integrity of an α-helix is in part dependent on correct steric configuration. This regular pattern gives the α-helix very definite features with regards to the thickness of the coil and the length of each complete turn along the helix axis. Such a hydrogen bond is formed exactly every 4 amino acid residues, and every complete turn of the helix is only 3.6 amino acid residues. This coil is held together by hydrogen bonds between the oxygen of C=O on top coil and the hydrogen of N-H on the bottom coil. Note that R groups extend almost perpendicular from the axis.Īn α-helix is a right-handed coil of amino-acid residues on a polypeptide chain, typically ranging between 4 and 40 residues. Hydrogen bonds are shown as dotted bonds. Α-Helices Figure: Ball-and-stick model of the α helix. These structures are the first major steps in the folding of a polypeptide chain, and they establish important topological motifs that dictate subsequent tertiary structure and the ultimate function of the protein. The two most commonly encountered secondary structures of a polypeptide chain are α-helices and beta-pleated sheets. Secondary structure does not include bonding between the R-groups of amino acids, hydrophobic interactions, or other interactions associated with tertiary structure. Secondary structure refers to the shape of a folding protein due exclusively to hydrogen bonding between its backbone amide and carbonyl groups. The hydrogen bonding pattern in a section of a beta-strand is shown below. Loops are relatively disordered segments of protein chain, but often assume a very ordered structure when in contact with a second protein or a smaller organic compound.īoth helix and the beta-sheet structures are held together by very specific hydrogen-bonding interactions between the amide nitrogen on one amino acid and the carbonyl oxygen on another. The figure below shows two different views of the same beta-sheet: in the left-side view, the two regions of protein chain are differentiated by color. In a beta-sheet (or beta-strand) structure, two sections of protein chain are aligned side-by-side in an extended conformation. In a helix, the main chain of the protein adopts the shape of a clockwise spiral staircase, and the side chains point out laterally. For the most part, there are three important elements of secondary structure: helices, beta-sheets, and loops. Secondary structure is three-dimensional, but is a local phenomenon, confined to a relatively short stretch of amino acids. Below is a Lewis structure of a short segment of a protein with the sequence CHEM (cysteine - histidine - glutamate - methionine) A protein’s primary structure is two-dimensional - simply the sequence of amino acids in the peptide chain. Protein structure can be discussed at four distinct levels. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
